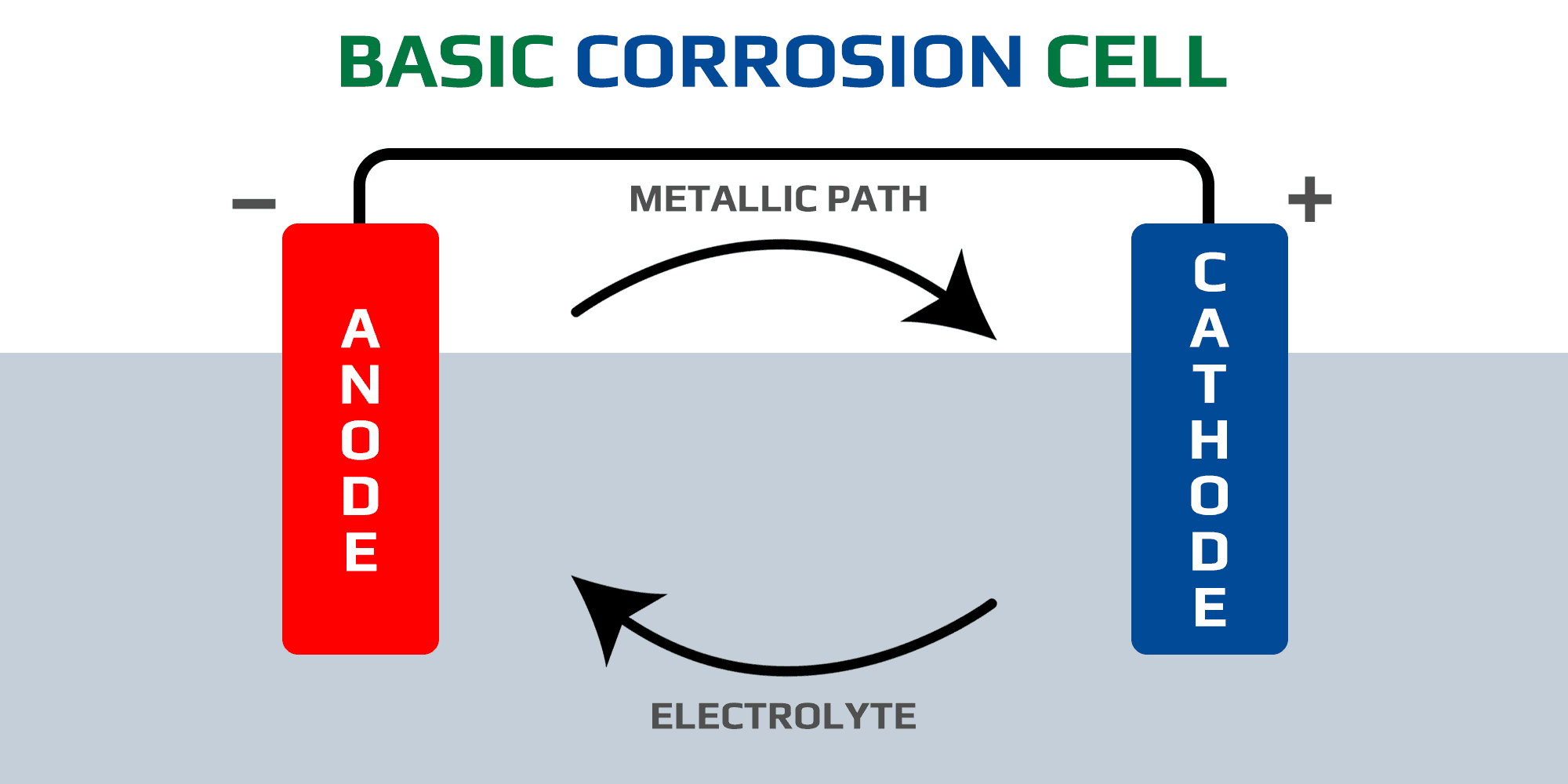
Corrosion is a silent killer in many industries, causing significant damage to equipment, infrastructure, and the environment. But did you know that understanding the basic corrosion cell is key to preventing or mitigating this degradation?
The basic corrosion cell consists of an anode, cathode, and metallic path. At the anode, metal atoms release electrons, which then flow through the metallic path to the cathode. Here, these electrons combine with oxidizing agents in the electrolyte, accelerating corrosion.
By grasping this fundamental mechanism, you can develop effective strategies to prevent or mitigate corrosion in your industry. At GreenVCI, we specialize in providing high-performance corrosion inhibitors to protect your assets from the ravages of corrosion.