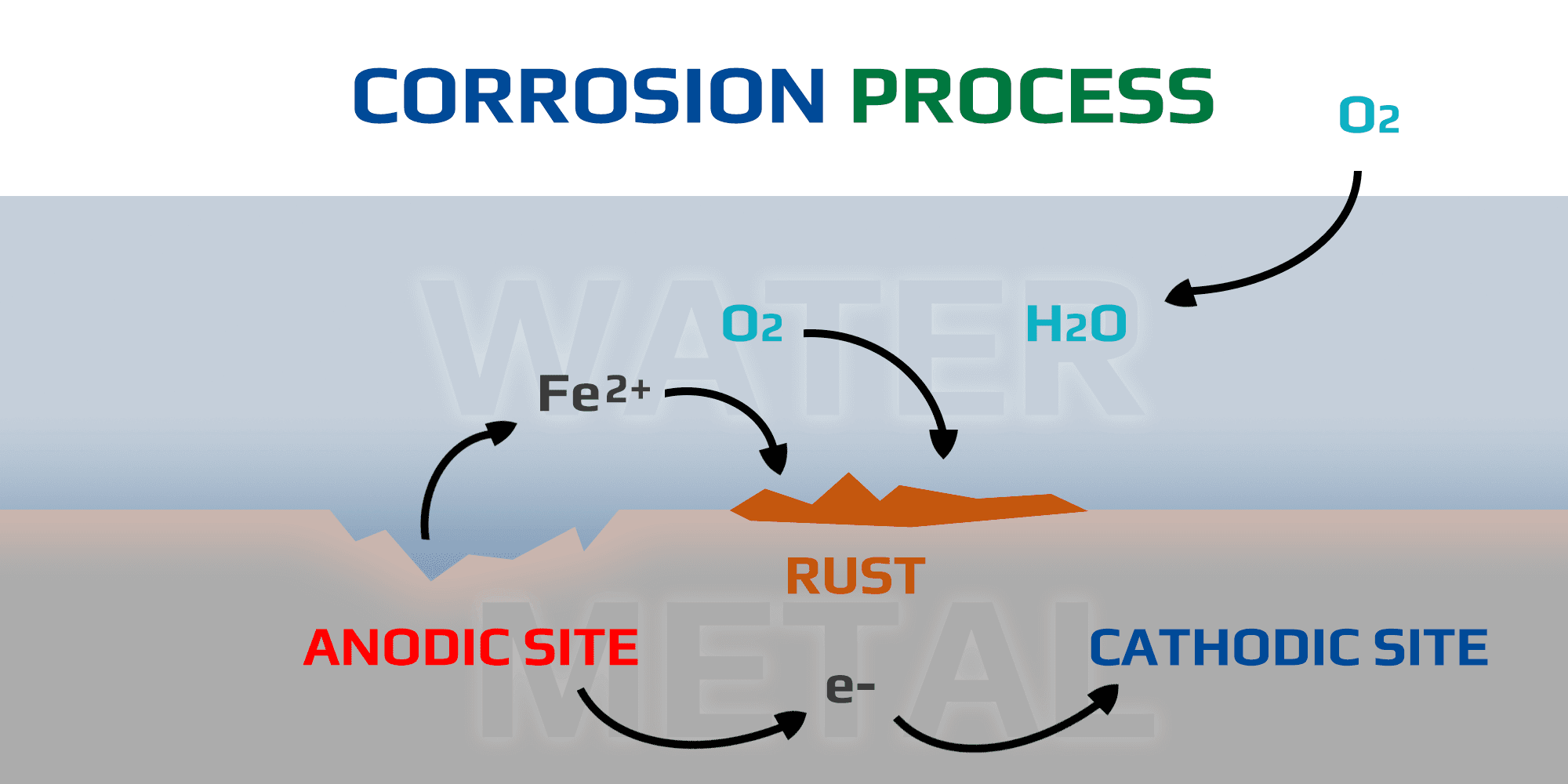
Corrosion is a complex process that can have devastating effects on equipment, infrastructure, and the environment. In this post, we’ll explore the corrosion process and how rust forms and spreads.
When iron metal undergoes oxidation reactions at anodic sites, it loses electrons to its surroundings at cathodic sites, producing ferrous ions (Fe2+). These ions then react with water and oxygen to form hydrated iron oxides, commonly known as rust. The presence of ferrous ions facilitates further corrosion reactions, leading to the continued breakdown of the metal surface.
As corrosion progresses, more ferrous ions are released into the solution, perpetuating the cycle of oxidation and reduction. The accumulation of rust and the continuous generation of ferrous ions contribute to the ongoing degradation of the metal structure over time.